Documentation Index
Fetch the complete documentation index at: https://docs.revilico.bio/llms.txt
Use this file to discover all available pages before exploring further.
Why Use this product?
Solubility is a core property that should be computationally tested in a way that represents biological and lab based conditions for downstream experimentation. In later lead optimization stages, many compounds may fail due to bad solubility in certain solvent conditions which has potential to cause downstream issues with bioavailability, assay reliability, and formulations. Revilico’s Solubility Engine allows chemists to test a compound’s intrinsic solubility across temperature gradients and different solvents, quickly and effectively.
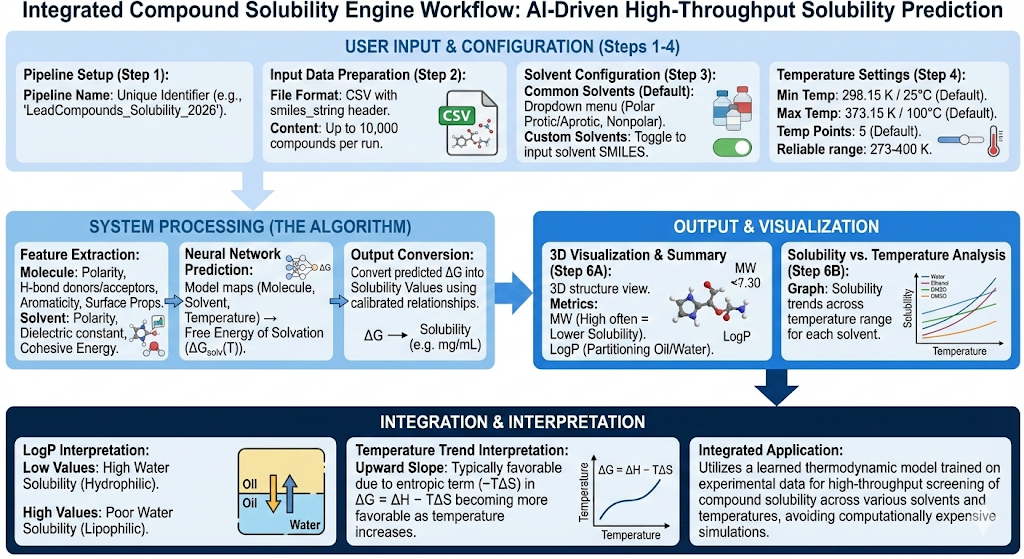
Background
Compound solubility is an important principle for evaluating the effectiveness of the delivery of a small molecule, as the molecule must be sufficiently dissolved to be absorbed, distributed and tested reliably. Poor solubility can limit bioavailability, cause assay failures, complicate formulation, and ultimately prevent an otherwise potent compound from becoming a viable drug. Revilico’s Compound Solubility Engine proves to be an effective tool for a high throughput screening of several molecules particularly measuring for solubility, saving both time and money for downstream experimentation.
Diving down into the mathematical theory behind solubility prediction, the binding free energy of solvation as a function of temperature can be defined by the following equation:
ΔG(T)=ΔH−TΔS
Where (Delta H) is the enthalpic term which is the net energy change resulting from breaking and forming of chemical interactions (i.e. bonds, intermolecular forces) and (Delta S) is the entropic term which measures the shift in molecular disorder, randomness, or energy dispersal during a physical or chemical process. In order for us to calculate this property across several compounds, it can become computationally intensive. This would require us to run molecular dynamics and free energy calculation engines at scale, and due to high computational cost, it is infeasible for speedy on-demand screening. However, alternative deep learning models can be utilized to calculate these properties at scale across several different conditions.
Now diving into the workflow behind this engine, computing solubility from first principles will be a very difficult task where we will need to model the solid crystal lattice, model the solvent explicitly, simulate molecules leaving the crystal, and sample many configurations, a task that would be very computationally expensive. Instead we will measure solubility using a learned thermodynamic model that is trained on experimental solubility data. Essentially the mapping of this model to predicted solubility free energy can be defined by the following equation:
(molecule,solvent,T)→ΔGsolv(T)
This model would take our molecule and extract out molecular size and shape, polarity, hydrogen bond donor/acceptors, aromaticity, flexibility, approximate charge distribution, and whether it is a hydrophobic vs hydrophilic surface through a series of “features” that will be fed into a neural network. Utilizing this data and the molecular featurizations we create, we aim to understand the general ability for the compound to dissolve in its subsequent solvents.
The model will then take the solvent and extract out polarity, dielectric constant, hydrogen bonding ability, and cohesive energy to ensure molecular features that are extracted will match well with certain solvent features.
After encoding both our molecule and solvent, we will then encode temperature, apply a trained neural network to predict our (Delta G) of solvation, then convert this (Delta G) to solubility as a result of the learned or calibrated relationships based on the data.
Interactive Results Viewer
Explore compound solubility predictions interactively. Select a molecule to view its predicted solubility across different solvents and temperatures.


