Documentation Index
Fetch the complete documentation index at: https://docs.revilico.bio/llms.txt
Use this file to discover all available pages before exploring further.
The Problem You Are Trying to Solve
“I have a set of lead compounds, and I want to plan practical synthetic routes and pathways so I can prioritize what to make next.”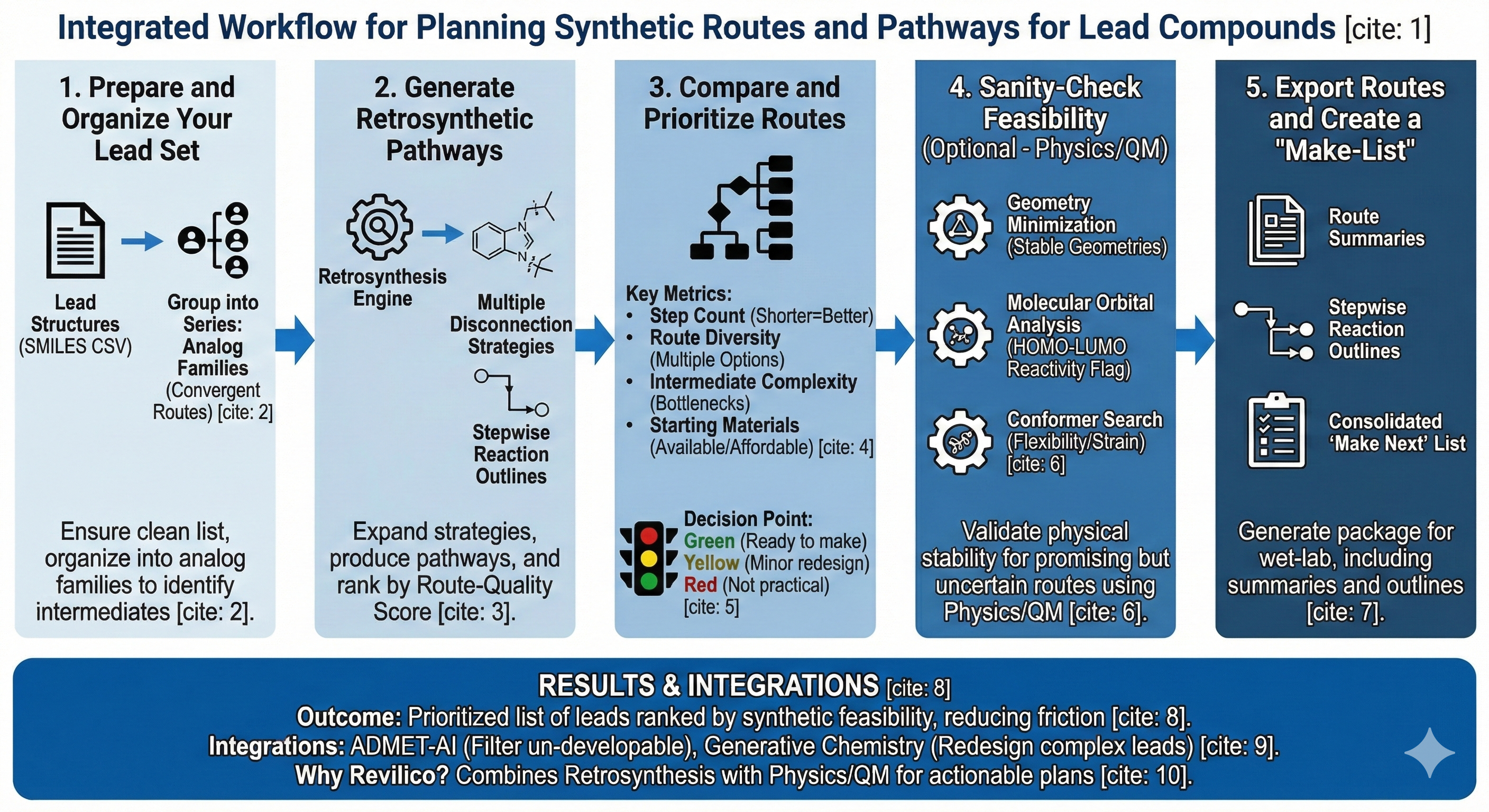
- Limited starting material availability
- Route length, yield, and step complexity
- Risky transformations or fragile intermediates
- Cost, cycle time, and scalability considerations
Solution
This workflow uses Revilico’s Retrosynthesis engine as the core route-planning layer, supported by optional feasibility and risk checks from other chemistry engines. The primary synthetic planning chain is: Lead Set Preparation → Retrosynthesis Route Generation → Route Ranking & Comparison → Starting Material & Risk Review → Export + Iterate. Binding, property, and AI engines can be integrated to ensure you prioritize the compounds that are both valuable and makeable.What Data Do I Need to Provide?
Required- Lead structures as SMILES (CSV upload or manual input)
- Optional compound identifiers (name / series / project tag)
- Any “hard constraints” from your chem team (must avoid certain reagents, protect certain groups, limit step count, etc.)
- Known preferred intermediates or supplier catalogs (if your org has them)
- Target number of steps / cost ceilings / timeline constraints
Workflow
- Prepare and Organize Your Lead Set
- Upload a CSV of lead SMILES (plus optional IDs/names)
- Confirm structures are valid and standardized
- Group compounds into series (if relevant) so you can compare routes across analog families
- Generate Retrosynthetic Pathways
- Expand multiple disconnection strategies per molecule
- Produce stepwise pathways with intermediates and reaction class labels
- Rank routes by a route-quality score (route plausibility, efficiency, starting material reasonableness)
- How would I make this?
- How many routes exist?
- Which ones look most realistic?
- Compare and Prioritize Routes Across Leads
- Step count (shorter is usually faster and lower risk)
- Route diversity (multiple independent options reduces project fragility)
- Intermediate complexity (risk of bottlenecks)
- Starting material practicality (availability and cost proxies)
- Convergence opportunities (shared intermediates across a series)
- At this point, all of the data can be sent to your chemistry team for utilizing these newly generated hypotheses as a baseline for getting these molecules synthesized
- Ready to make now
- Worth minor redesign to reduce synthesis complexity
- Not currently practical relative to alternatives
- Sanity-Check Molecular Feasibility and Stability (Optional)
Optional supporting engines:
- Geometry Minimization and Thermochemistry to sanity-check stable geometries and identify strained or unstable candidates
- Molecular Orbital Analysis (HOMO–LUMO) to flag potentially reactive or unstable electronic profiles (helpful for identifying “looks good, but might be chemically problematic” cases)
- Conformer Search to highlight extreme flexibility or conformational strain that could complicate synthesis or isolation
- Export Routes and Create a Make-List
- Route summaries per compound
- Stepwise reaction outlines and intermediates
- A consolidated “Make Next” list for your synthesis team
- With the data on hand for planning synthesis, your chemists can now move forward with getting the molecules synthesized.
- If you are using this engine as a secondary screen to any other engine, you can utilize the results as a sanity check of the compounds to ensure that the compounds made with generative chemistry are feasible to move forward with.
Integration with Other Engines (Optional)
Synthetic planning rarely happens in isolation. Revilico supports tight integration with:- ADMET-AI + Solubility to avoid planning routes for compounds likely to fail developability
- Docking / MD / FEP to ensure synthesis effort is directed toward leads with strong on-target justification
- Generative Chemistry to redesign hard-to-make leads into more synthesizable analogs while preserving activity motifs
Why Revilico?
Revilico makes synthetic planning actionable by combining:- A dedicated retrosynthesis engine for route generation and ranking
- Optional physics/QM checks for stability and feasibility confidence
- AI-assisted interpretation to speed prioritization and iteration
- Integrations with design, screening, and optimization workflows so you can plan synthesis for the right compounds, not just the most interesting ones on paper.

